Part 40: Closed Tournament - Round 5
Closed Tournament - Round 5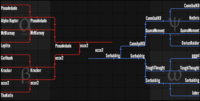
Congratulations to ecco2! You do not have to do anything this week except laugh at the poor fools below.
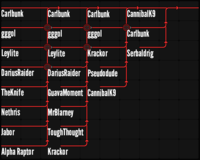
LOSER'S BRACKET
So I found this one puzzle by Lyosha that was horribly designed. It was basically one where you had no way to separate the two random inputs (differently-bonded carbon groupings) unless you performed some fusion/fission guesswork and then stored the waste every time you guessed wrong. I was pretty annoyed by the fact that there was no good way to run it without a state machine.
However, I came across something one day when reading about chemical chirality. In fact, I believe Carbon dioxide talked about it at one point.
Chemists noticed that tartaric acid derived from organic sources (in this case, wine lees) would polarize light passing through it. A weird thing, though, was that tartaric acid derived through chemical synthesis did not polarize light.
As it turns out, Louis Pasteur caught something that previous chemists had overlooked: the tartaric acid crystals in the synthetic version were shaped slightly differently when viewed through a microscope. Something like this:
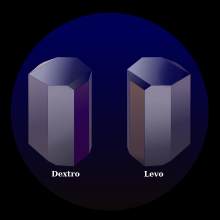
It turns out that the synthetic tartaric acid had roughly equal amounts of either crystal, canceling the polarization out. By sorting these crystals manually, he was able to produce a pure sample of levotartaric acid. Boy, that must have been a pain.
Anyway, I realized something incredible: what if Lyosha was a modern day Pasteur? What if this was actually just the ultimate state machine challenge in disguise?
Isomer sorting
by Lyosha

Task: Produce as many carbon isomers as you can before the reactor crashes/stops outputting.
Goal Priority: MOST OUTPUTS/LEAST CYCLES/LEAST SYMBOLS/SUBMISSION TIME
H4sIAIcQT1IA/62QsWrDMBCGXyXcbIMkgwd7zNJC106lg+qcY4MiGek0uMbv3pOVQNwEutTDgT
/d/fpOC4x2ilR+O4sBmgVEKhvj348FLs5gFw1CA0ftv5w9vLfHVqlailbJWshWiVSrXDdSqVoI
KKBz0RI09Vo8y3lJOduUSv0pTV5nbznp9D7ncy1A/iXIsVLsBKTiSR51kR53fS5WPayW9bLqrw
Wl4O+m9g8PlvM4cNChNNqfsczq0PTaBCyAY0/oy2u/yJ0BbXAeGvIRM+ljwB0IkxmJ9ozQ4OT8
HaV5SvoeA2rfDSxm9SWR1+Au6A98C432zFxHGtKV8Da7MGgmp7HvR96fZvZafwAFsP9dXwIAAA
==
If you would like to read more about Louis Pasteur's discovery, you can do so here.